|
12/27/2023 0 Comments Heisenberg principle equation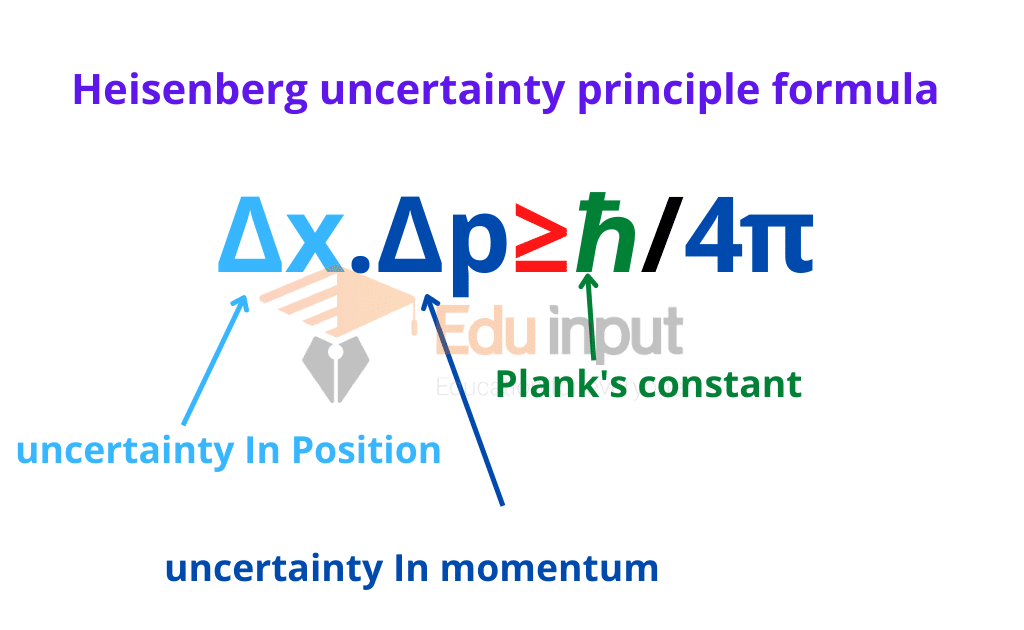
Now, apply the Heisenberg’s Principle to an electron in an orbit of an atom, with h = 6.626 × 10⁻³⁴ Js and m= 9.11 × 10⁻³¹ Kg, Since momentum, p=mv, by this Heisenberg’s uncertainty principle formula can also be written as:Īccurate measurement of position or momentum automatically indicates a larger error in the measurement of the other quantity. Therefore, from this concept we can write: In this principle, ∆x is considered to be an error in the position measurement whereas ∆p is the error in momentum. So, an accurate measurement of one quantity leads to large uncertainty in the measurement of the other.ĭerivation of the Heisenberg Uncertainty Principle Whereas the particle having a well-defined wavelength gives the precise velocity. This shows that particles which have definite position have no certain or fixed velocity. The more the undulation of the particle, the more ill-defined becomes the wavelength, which helps in the determination of momentum of the particle. As each particle has a wave nature, the probability of finding particles is maximum where the undulations of the wave are greatest. The main reason behind the origin of the uncertainty principle is the dual nature of a wave-particle. Here ∆ denotes the uncertainty in that variable and h is the Planck's constant and its value is 6.6 × 10 −34 joule-second. Heisenberg principle can be numerically written as: ∆X × ∆p ≥ h/4π From this concept it is clear that uncertainties in the conjugate pairs of momentum/position and energy/time were termed by Heisenberg as having a minimum value corresponding to the Planck’s constant divided by 4π. The same relationship can also be applicable for energy and time, as we cannot measure the exact energy in a given finite amount of time. 
This principle works in contrast to classical Newtonian physics which states all variables of particles to be measurable to an arbitrary uncertainty given good enough equipment.Īs per the uncertainty principle if you know the exact position of a particle it is impossible to determine the exact momentum of the particle and vice versa. However, this principle mainly focuses on position and momentum of particles, the principle states that the more precisely the position is known the more uncertain the momentum is and vice versa. This principle states that there is inherent uncertainty in the act of measuring a different given variable of particles. This principle is one of the most celebrated results of quantum mechanics and it states that we can determine different things about particles simultaneously at a time. This principle was stated by the German physicist Werner Heisenberg in the year 1927. The uncertainty principle is called the Heisenberg uncertainty principle.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
